by Jacob Bell – BioPharmaDive –
The agency has now cleared Shanghai-based Unixell to begin human studies in the U.S. for an allogeneic, stem cell-derived therapy targeting focal epilepsy.
For the first time, the Food and Drug Administration is allowing a certain kind of cell therapy for epilepsy to be tested in humans.
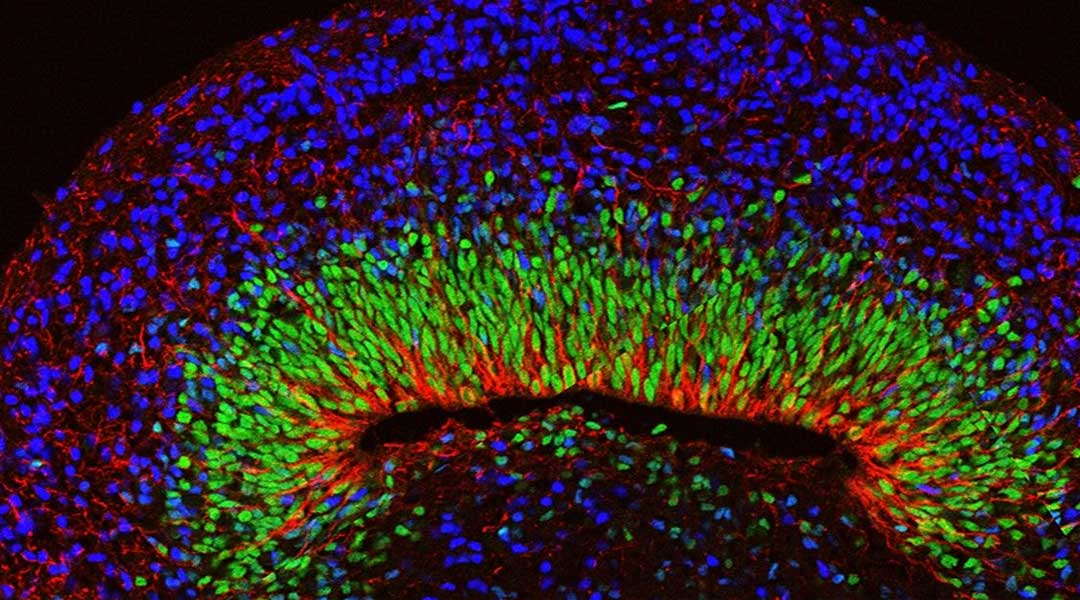
The therapy, created by Shanghai-based Unixell Biotechnology, is designed to curb the excessive electrical activity that triggers seizures in epileptic patients. It uses donor-derived — or “allogeneic” — stem cells reprogrammed so that they ultimately produce the main chemical messenger, “GABA,” responsible for calming the brain and nervous system.